
Although there is helium on Earth, it was first discovered by observing the sun.The country Argentina is named after the element silver (symbol Ag) which is argentum in Latin.The only letter not in the periodic table is the letter J.There are probably no more than a few ounces of it on earth at any given time. These trends became apparent to Russian chemist Dmitri Mendeleev (18341907) when he arranged the elements in a table in order of increasing mass. Francium is the rarest element on earth. The Periodic Table of the Elements 23.0 Na sodium 11 39.1 K potassium 19 40.1 Ca calcium 20 45.0 Sc scandium 21 47.9 Ti titanium 22 50.9 V vanadium 23 52.0 Cr chromium 24 54.9 Mn manganese 25 55.8 Fe iron 26 58.9 Co cobalt 27 58.7 Ni nickel 28 63.5 Cu copper 29 65.4 Zn zinc 30 69.7 Ga gallium 31 72.6 Ge germanium 32 74. Periodicity refers to the recurring trends that are seen in the element properties.The s s sublevel has one orbital, the p p sublevel has three orbitals, the d d sublevel has five orbitals, and the f f sublevel has seven orbitals. Recall that the four different sublevels each consist of a different number of orbitals. :max_bytes(150000):strip_icc()/periodictable-56a129413df78cf77267f8df.jpg)
Carbon is important to the existence of life. 1: Period Length and Sublevels in the Periodic Table. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. A new period begins when a new principal energy level begins filling with electrons. 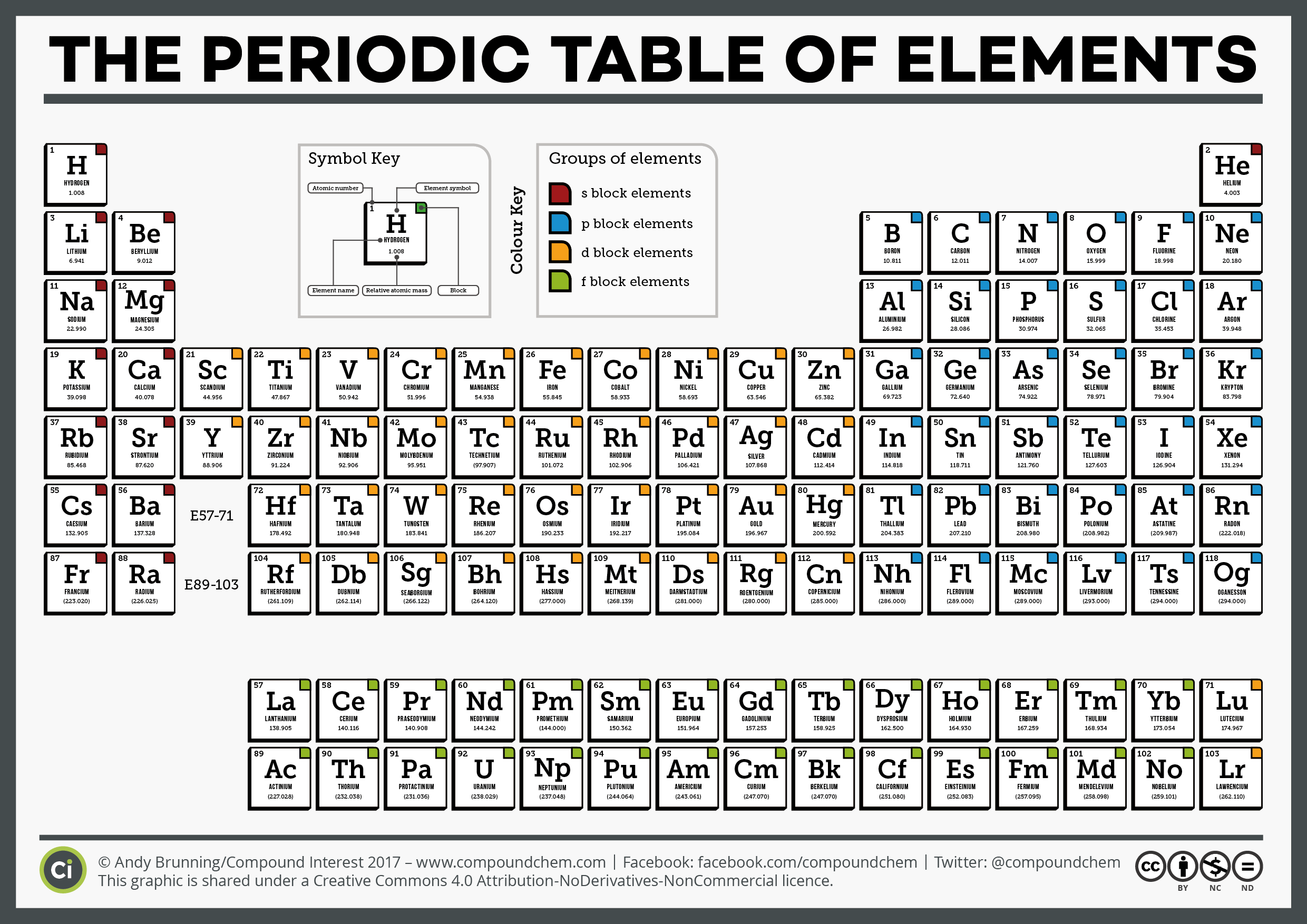
There are seven periods in the periodic table, with each one beginning at the far left. Carbon is unique in that it is known to form up to 10 million different compounds. The modern periodic table is based on Dmitri Mendeleev’s 1896 observations that chemical elements can be grouped according to chemical properties they exhibit. A period is a horizontal row of the periodic table.Using the table, Mendeleev was able to accurately predict the properties of many elements before they were actually discovered. The periodic table was proposed by Russian chemist Dmitri Mendeleev in 1869. For gold the "Au" comes from the Latin word for gold "aurum". Some are a bit harder like Fe for iron or Au for gold. Some of the abbreviations are easy to remember, like H for hydrogen. They can understand and predict how an element might react or behave in a certain situation.Įach element has its own name and abbreviation in the periodic table. This lining-up and grouping of similar elements helps chemists when working with elements. You can see all the groups in the table below. A table of the chemical elements arranged in order of atomic number, usually in rows, so that elements with similar atomic structure (and hence similar chemical properties) appear in vertical columns. They are all very similar in that they have only 1 electron in their outer shell and are very reactive. Another example is the alkali metals which all align on the left-most column. Element atomic number increases moving from left to right across a period. They all have a full outer shell of electrons, making them very stable (they tend not to react with other elements). There are seven rows of the periodic table, which are called periods. These elements all line up in the eighteenth or last column of the periodic table. \) (Credit: Christopher Auyeung and Joy Sheng Source: CK-12 Foundation License: CC BY-NC 3.One example of a group is the noble or inert gases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
